
Organic Chemistry,6th Edition L.G.Wade,Jr. Chapter 22 Alpha Substitution and Condensations of Enols and Enolate lons Jo Blackburn Richland College,Dallas,TX Dallas County Community College District ©2006,Prentice Hall
Chapter 22 Alpha Substitution and Condensations of Enols and Enolate Ions Jo Blackburn Richland College, Dallas, TX Dallas County Community College District © 2006, Prentice Hall Organic Chemistry, 6th Edition L. G. Wade, Jr

Alpha Substitution Replacement of a hydrogen on the carbon adjacent to the carbonyl,C=O. Step 1:Deprotonation Step 2:Attack on electrophile base: enolate ion Copyright 2005 Pearson Prentice Hall,Inc. > Chapter 22 2
Chapter 22 2 Alpha Substitution Replacement of a hydrogen on the carbon adjacent to the carbonyl, C=O. =>
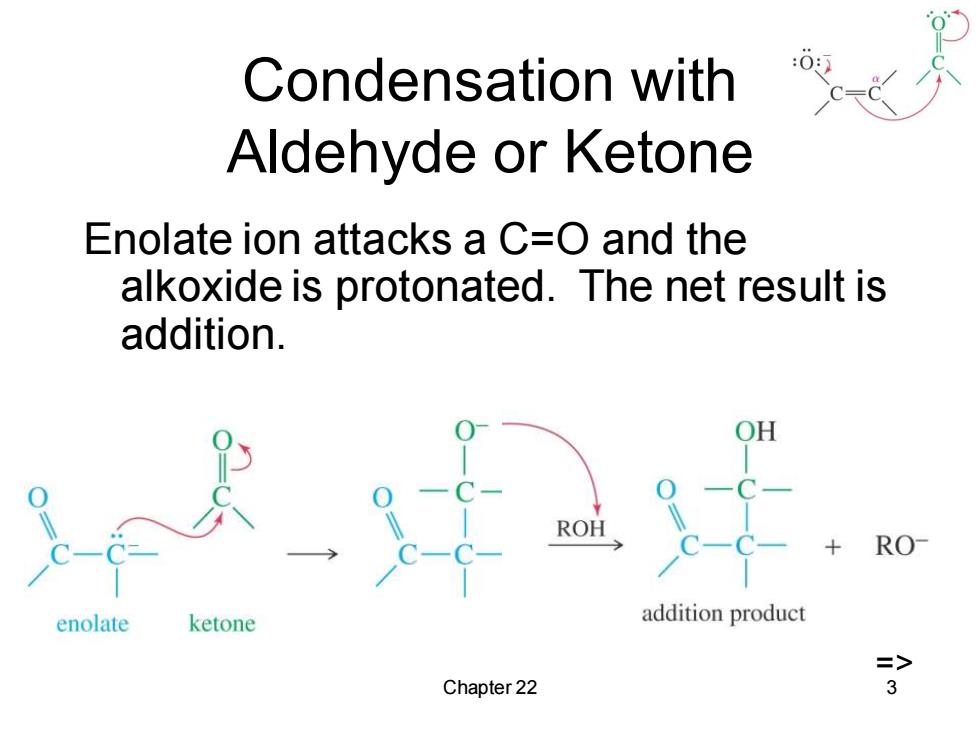
Condensation with Aldehyde or Ketone Enolate ion attacks a C=O and the alkoxide is protonated.The net result is addition. RO enolate ketone addition product => Chapter 22 3
Chapter 22 3 Condensation with Aldehyde or Ketone Enolate ion attacks a C=O and the alkoxide is protonated. The net result is addition. =>
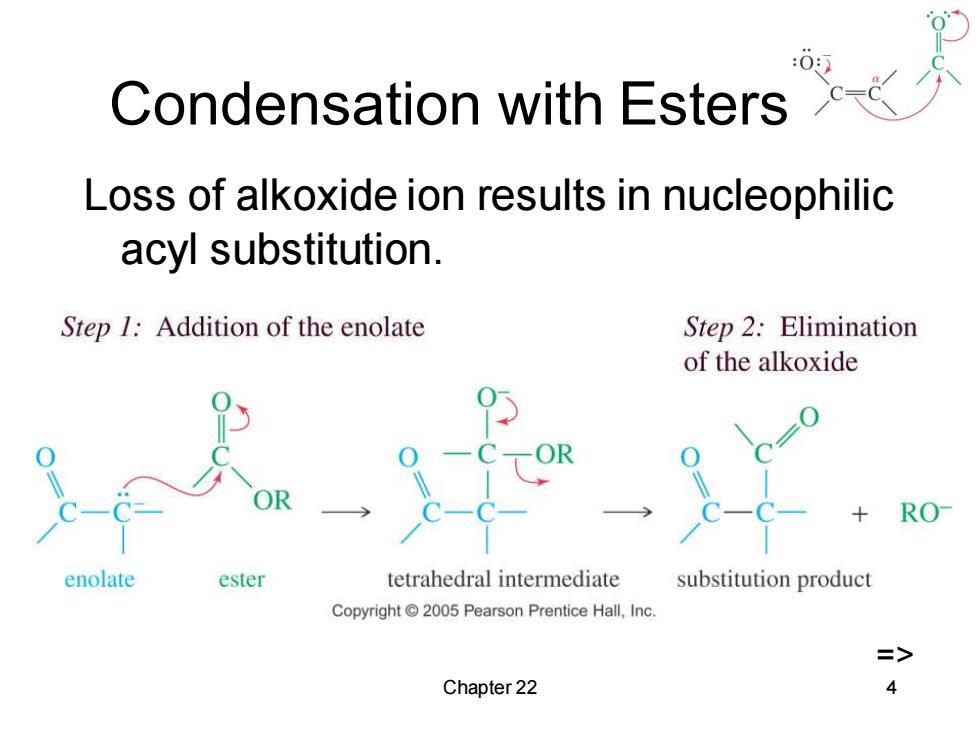
Condensation with Esters Loss of alkoxide ion results in nucleophilic acyl substitution. Step 1:Addition of the enolate Step 2:Elimination of the alkoxide RO enolate ester tetrahedral intermediate substitution product Copyright 2005 Pearson Prentice Hall,Inc. => Chapter 22 4
Chapter 22 4 Condensation with Esters Loss of alkoxide ion results in nucleophilic acyl substitution. =>
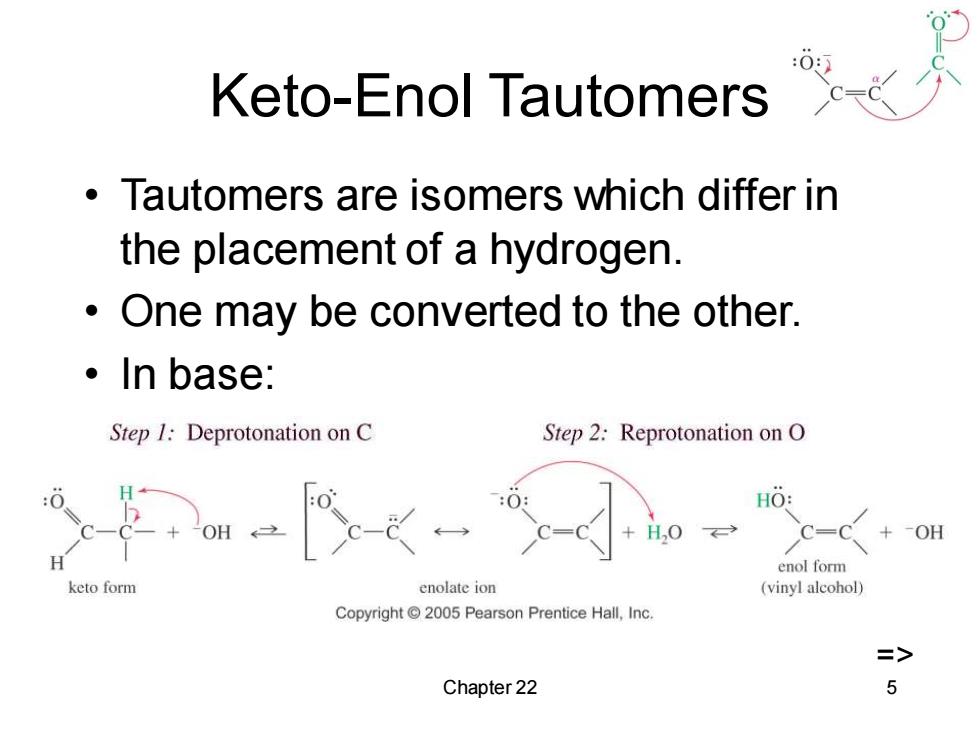
Keto-Enol Tautomers Tautomers are isomers which differ in the placement of a hydrogen. One may be converted to the other. ·In base: Step 1:Deprotonation on C Step 2:Reprotonation on O HO: OH +OH H enol form keto form enolate ion (vinyl alcohol) Copyright2005 Pearson Prentice Hall,Inc. => Chapter 22 5
Chapter 22 5 Keto-Enol Tautomers • Tautomers are isomers which differ in the placement of a hydrogen. • One may be converted to the other. • In base: =>